Avian
parasites study group


Ph. D. students:
Magdalena Fialová
Anna Brotánková
Ashwin Kumar
Master students:
Anna Kadlecová
Lenka Geržová
Aneta Karafiátová
Bachelors:
Alumni:
Jan Votýpka, Lenka Zídková
Ondřej Černý, Barbora Chalupová, Anežka Santolíková, Petr Synek, Jana Szabová
Kristýna Dulavová, Kristýna Chobotová, Lada Janíčková, Anna Filousová, Iveta Sekerášová
UNIGOU student: Ariane Rampazzo
in cooperation with
Ivan Čepička (Department of Zoology) WWW
Kristýna Hrazdilová (Masaryk University, Brno)
Ana Rivero (University of Montpellier)
Miroslav Šálek (University of Life Sciences)
Petr Voříšek (Czech Society for Ornithology)
Karel Weidinger (Palacky University, Olomouc)
We study vector-parasite-host interactions in bird populations.
The particular ai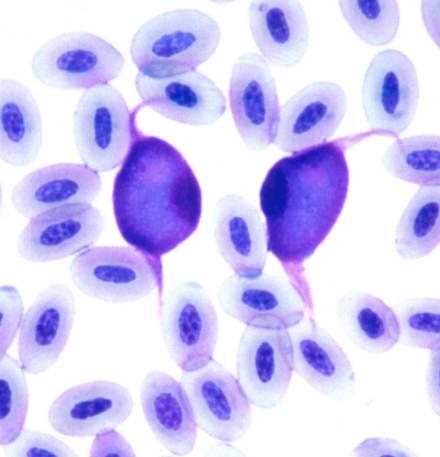 ms are:
ms are:
* Identification of blood parasites, their prevalences and modes of transmission
* Seasonal dynamics of ornithophilic Diptera
* Trypanosome host specificity, life cycles in the vectors and experimental transmission
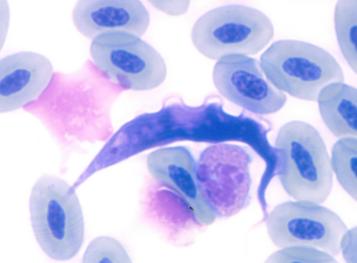
* Biodiversity of avian trypanosomes and haemosporidia
We have a collection of 450 strains of avian trypanosomes isolated from passerines, 200 from raptors, and 150 from bloodsucking vectors; and blood collected from 10000 individuals of 50 passerine species.
In an ongoing project, we plan to compare the information obtained from bloodmeal barcoding and parasite lineages present in the vectors to assess host preferences of bloodsucking insects.
We study the influence of trypanosomes on mosquito fitness.
Tits (Parus s. l.) and warblers (Acrocephalus spp.) are our model hosts for the study of blood parasites community and the dynamics and coinfections, including trypanosomes, haemosporidians, and lankesterellids.
Oustanding results:
Lankesterella is a genus of blood coccidians thas has been neglected in birds for a long time. We show that these parasites have a considerable degree of host specificity, probably at the level of host genus. Besides Lankesterella species, Isospora parasites can be readily detected in blood of passerines; barcoding of positive cases is thus essential (Venkatachalam et al. 2023).
Persistence of vector-transmitted parasites in vertebrate hosts is crucial fot their survival to the next transmission season. We have studied the diversity and persistence of three genera of sparrowhawk parasites. Prevalences increased with age but in some cases, hidden turnover of lineages occurred (Svobodová et al. 2023).
The wide ability of trypanosomes to use diverse vectors was confirmed again in our study of louse flies (Hippoboscidae) and avian trypanosomes. Three species were confirmed as new vectors, and louse flies were shown to harbour several trypanosome lineages, with intercontinental occurrence. Migratory and resident birds host different species of louse flies (Santolíková et al. 2022).
Vectorial plasticity of Megatrypanum trypanosomes is unexpectedly high, as revealed by our studies of mammalian T. theileri. Related to avian trypanosomes, they use mosquitoes as vectors besides a wide range of Diptera. The prevalence in Aedes species can exceed 25%; the isolates thrive in laboratory mosquitoes after experimental infection. As avian trypanosomes, T. theileri infectious forms are expelled in urine while the mosquito is feeding, enabling thus tranmsission via conjunctiva (Brotánková et al. 2022).
Mosquitoes were among the first insects found to harbour trypanosomes; however, their role in transmission was confirmed only recently for T. culicavium. We described the complete life cycle of yet another mosquito trypanosome, T. thomasbancrofti, which resides in the hindgut of mosquitoes, and is related to blackfly-transmitted T. avium (Fialová et al. 2021).
Avian trypanosomes display a high plasticity in the use of dipteran vectors. Recently, we have confirmed the potential of phlebotomine sandflies to transmit avian trypanosomes (Svobodová and Rádrová 2018), which has been earlier suggested by findings of Lutzomyia longipalpis naturally infected with T. avium. Sandflies should be added to the list of avian trypanosomes vectors.
Life cycle of trypanosomes from T. bennetti group was elucidated using laboratory-bred
model hosts, midge Culicoides nubeculosus and the canary, and confirmed by discovery of
naturally infected vectors - ornithophilic biting midges. Biting midges are highly susceptible to artificial infection, and can be infected after feeding on T.bennetti-infected bird. Transmission probably occurs by ingestion of the vector (Svobodová et al. 2017).
Ornithophilic bloodsucking Diptera (Culicidae, Simuliidae, Hippoboscidae) were microscopically examined for the presence of trypanosomatids in their guts. We dissected 3270 specimens, namely Culex pipiens (n=898), C. modestus (136), Simulium vernum (1455), S. angustipes (221), and Ornithomyia avicularia (524). All insect species have been found infected with trypanosomatids, and the prevalence usually ranged between 4 to 8% but reached 60% in S. vernum. Blackflies and hippoboscids harboured exclusively trypanosomes (both T. cf. avium s.s.; T.corvi/culicavium group in hippoboscids). Mosquitoes were infected with T. culicavium and T. avium s. l. but also with monoxenous parasites including Paratrypanosoma confusum, which was established in culture. Only 4% of the isolated parasite strains were monoxenous while the majority were avian trypanosomes, confirming the vectorial status of the studied insects (Svobodová et al., 2015a).
The role of shared vectors and parasite phylogenetic relationships on the occurrence of haemoparasites in the nesting populations of raptors in two different areas of the Czech Republic was studied. Altogether, 788 nestlings and 258 adult Eurasian sparrowhawks (Accipiter nisus) and 321 nestlings and 86 adult common buzzards (Buteo buteo) were screened for Trypanosoma, Leucocytozoon, and Haemoproteus. We conclude that phylogenetically unrelated parasites that share the same vectors tend to have similar distributions within the host populations of two different raptor species. (Svobodová et al. 2015b).
In birds and vectors, we have found at least 12 lineages of avian trypanosomes, which split into 3 different groups according to the SSU rRNA gene and RAPD analysis. Each group was further subdivided into several lineages, some of them representing already described or putatively distinct species of avian trypanosomes. Only some lineages included isolates from both avian and vector hosts while many consisted only from bird or insect host isolates (Zídková et al. 2012)
We have described a new trypanosome species, T. culicavium, isolated from culicines. Its transmission was achieved using laboratory-bred Culex quinquefasciatus and canaries. Although the occurrence of trypanosomes on the stomodeal valve suggested transmission by bite (Volf et al. 2004), we have demonstrated that it occurs by ingestion. Natural avian host was found among mosquito-eating passerines (Votýpka et al. 2012).
Different height preferences of ornithophilic vectors were demonstrated using CDC traps baited with chickens.Culicine mosquitoes occur at ground while blackflies in canopy level (Černý et al. 2011). This probably influences potential host spectra of transmitted trypanosomes (T. culicavium, T. cf. avium), and is in accordance with respective modes of transmission, namely ingestion by mosquito-eating birds in T. culicavium, and prediuresis into conjunctiva in T. cf. avium from blackflies to raptors.
Ornithophilic insects have been studied in nestboxes. While midges were caught readily, blackflies and mosquitoes were almost absent (Votýpka et al. 2008). Trypanosomes previously reported in nestlings of cavity breeders thus should not belong to T. cf avium, since it is transmitted by blackflies, but rather to T. bennetti, T. culicavium or other species of avian trypanosomes.
By comparing SSU rDNA sequences of raptor and bloodsucking insects isolates, we have confirmed that T. cf. avium is transmitted by blackflies (Votýpka et al. 2002). Infective forms isolated directly from blackflies (Eusimulim spp.) caught in the wild were used to show that transmission occurs by ingestion or prediuresis (Svobodová & Votýpka 2004).
We have isolated several monoxenous kinetoplastids from bloodsucking Diptera. Three new species of monoxenous kinetoplastids were described from biting midges: Herpetomonas ztiplika, H. trimorpha, and Sergeia podlipaevi (Podlipaev et al. 2004, Zídková et al. 2010, Svobodová et al. 2007). Their transmission to laboratory-bred biting midges was possible from sugar.
Manipulation of host behaviour is a common strategy of trophicaly transmitted parasites which causes increased predation risk. We showed that voles infected with Sarcocystis spp. are more often preyed upon, To exlude the sampling bias (traps vs. birds nests), we demostrated the same experimentally using laboratory mice. Rodents infected with Sarcocystis spp. with birds of prey as definitive hosts have three times higher probability of becoming a prey (Voříšek at al. 1998).
Publications
Venkatachalam AKSB; Čepička I; Hrazdilová K; Svobodová M (2023) Host specificity of passerine Lankesterella (Apicomplexa: Coccidia). European Journal of Protistology 90, 126007 PDF
Svobodová M, Čepička I, Zídková L, Kassahun A, Votýpka J, Peške L, Hrazdilová K, Brzoňová J, Voříšek P, Weidinger K (2023) Blood parasites (Trypanosoma, Leucocytozoon, Haemoproteus) in Eurasian Sparrowhawk (Accipiter nisus): Diversity, incidence, and persistence of infection at the individual level.
Parasites and Vectors PDF
Santolíková A, Brzoňová J, Čepička I, Svobodová M (2022) Avian Louse Flies and Their Trypanosomes: New Vectors, New Lineages and Host–Parasite Associations. Microorganisms 2022, 10, 584 (PDF)
Brotánková A, Fialová M, Čepička I, Brzoňová J, Svobodová M (2022) Trypanosomes of the Trypanosoma theileri Group: Phylogeny and New Potential Vectors. Microorganisms 2022, 10, 294; https://doi.org/10.3390/microorganisms10020294 (PDF)
Fialová M, Santolíková A, Brotánková A, Brzoňová J, Svobodová M (2021) Complete life cycle of Trypanosoma thomasbancrofti, an avian trypanosome transmitted by culicine mosquitoes. Microorganisms 2021, 9, 2101 https://doi.org/10.3390/microorganisms9102101 (PDF)
Svobodova M; Radrova J (2018) Phlebotomine sandflies - Potential vectors of avian trypanosomes. Acta Protozoologica 57: 53-59 PDF
Svobodová M, Dolnik OV, Čepička I, Rádrová J (2017) Biting midges (Ceratopogonidae) as vectors of avian trypanosomes. Parasites and Vectors 10: 224 PDF
Svobodová M, Volf P, Votýpka J. (2015) Trypanosomatids in ornithophilic bloodsucking Diptera. Medical and Veterinary Entomology 29: 444-447 PDF
Svobodová
M, Weidinger K, Peške L, Volf P, Votýpka J, Voříšek P. (2014) Trypanosomes and haemosporidia in the buzzard (Buteo buteo) and sparrowhawk (Accipiter nisus): factors affecting the prevalence of parasites. Parasitology Research 114: 551-560 PDF
Kristofik J; Darolova A; Griggio M; Majtan J; Okuliarova M ; Zeman M ; Zidkova L; Hoi H. (2013) Does egg colouration signal female and egg quality in reed warbler (Acrocephalus scirpaceus)? (2013) Ethology Ecology & Evolution 25: 129-143 PDF
Zídková l., Szabová J., Čepička I., Svobodová M. (2012) Biodiversity of avian trypanosomes. Infection, Genetics and Evolution 12: 102-112 PDF
Valera F, Zídková L. Reproductive anatomy and fecundity estimation of the haematophagous ectoparasite Carnus hemapterus. Parasitol Research (2012) 110: 1733-1739 PDF
Votýpka J. , Szabová J. , Rádrová J., Zídková L. , Svobodová M. (2011) Trypanosoma culicavium sp. nov., an avian trypanosome transmitted by Culex mosquitoes. International Journal of Systematic and Evolutionary Microbiology 62: 745-754 PDF
Černý O., Votýpka J., Svobodová M. (2011) Spatial feeding preferences of ornithophilic mosquitoes, blackflies and biting midges. Medical and Veterinary Entomology 25: 104-108. PDF
Zídková L., Čepička I., Votýpka J., Svobodová M. (2010) Herpetomonas trimorpha sp. nov. (Trypanosomatidae, Kinetoplastida), a parasite of the biting midge Culicoides truncorum (Ceratopogonidae, Diptera). International Journal of Systematic and Evolutionary Microbiology 60: 2236-2246. PDF
Votýpka J., Synek P. & Svobodová M. (2009) Endophagy of biting midges attacking cavity-nesting birds. Medical and Veterinary Entomology 23: 277–280. PDF
Svobodová M, Zídková L, Čepicka I, Oborník M, Lukeš J, Votýpka J. (2007) Sergeia podlipaevi gen. nov., sp. nov. (Trypanosomatidae, Kinetoplastida): a parasite of the biting midges (Ceratopogonidae, Diptera). International Journal of Systematic and Evolutionary Microbiology 57:423-32. PDF
Votýpka J., Svobodová M. (2004) Trypanosoma avium: experimental transmission from black flies to canaries. Parasitology Research 92: 147-151 PDF
Modrý D., Votýpka J., Svobodová M. (2004) Note on the taxonomy of Frenkelia microti (Findlay & Midleton, 1934) (Apicomplexa: Sarcocystidae) Systematic Parasitology 58: 185-187 PDF
Svobodová M., Voříšek P., Votýpka J., Weidinger K. (2004) Heteroxenous coccidia (Apicomplexa: Sarcocystidae) in the populations of their final and intermediate hosts: European buzzard and small mammals. Acta Protozoologica 43: 251-260 PDF
Votýpka J., Oborník M., Volf P., Svobodová M., Lukeš J. (2002) Trypanosoma avium of raptors (Falconiformes): phylogeny and identification of vectors. Parasitology 125: 253-263 PDF
Voříšek P., Votýpka J., Zvára K., Svobodová M. (1998) Heteroxenous coccidia increase the predation risk of parasitized rodents. Parasitology 117: 521-524 PDF
Svobodová M., Votýpka
J. (1998) [Occurence of blood protists in raptors (Falconiformes)]. Buteo 10:
51-56 PDF
Svobodová M. (1997) [Heteroxenous
coccidia of raptors and owls from the Czech and Slovak Republics] Buteo 9: 35-42 PDF
Svobodová M. (1996) Sarcocystis from goshawk (Accipiter gentilis) with great tit (Parus major) as intermediate host. Acta Protozoologica 35: 223-226 PDF
Svobodová M., Cibulková M. (1995) Isospora sp. (Apicomplexa: Eimeriidae) of Icterine Warbler (Hippolais icterina, Passeriformes, Sylviidae): The possibility of parents to nestlings transmission. Acta Protozoologica 34: 233-235 PDF
Svobodová M. (1994) Isospora, Caryospora and Eimeria (Apicomplexa: Eimeriidae) of passeriform birds from Czech Republic. Acta Protozoologica 33: 101-108 PDF
Abstracts

Svobodová M, Dolnik OV, Čepička I, Rádrová J (2016) The role of biting midges in avian trypanosomes transmission. TryTax 2, 32
Rádrová J, Dolnik OV, Svobodová M (2016) The seasonal dynamics of avian trypanosomes in mosquitoes. TryTax 2, 55
Zídková L., Votýpka J., Čepička
I., Svobodová M. (2005) How many trypanosome species are there in birds? Alauda
73: 245
Černý O., Votýpka J., Svobodová M. (2005) Passerine trypanosomes: Morphological heterogeneity and spatial distribution of vectors. Alauda 73: 275-6
Votýpka J., Svobodová M., Volf P. (2002) Development of avian trypanosomes in mosquitoes. Journal of Eukaryotic Microbiology, http://www.jeukmic.org/abstr/int/c4/c410.html
Votýpka J., Svobodová M., Volf P., Lukeš J. (2001) Species composition and vectors of avian trypanosomes - molecular evidence. Journal of Eukaryotic Microbiology , www.jeukmic.org/abstr/int/c3/c308.html
Votýpka J., Svobodová M., Voříšek P., Peške L., Lacina D., Volf P. (2000) Blood parasites and haematophagous insects of raptors (Falconiformes) in the Czech Republic. Journal of Eukaryotic Microbiology 47(2): A http://www.jeukmic.org/abstr/int/c2/c208.html
Voříšek P., Diviš T., Svobodová M.: Distribution of Frenkelia parasites in the buzzard andits rodent prey. The Ornithological Notebook of the XXI International Ornithological Congress, Vienna, August 20-25, 1994
Svobodová M., Votýpka
J., Voříšek P. (1998): Frenkelia, buzzards and rodents: multiple mechanisms
of parasite survival strategy. Bulletin of the Scandinavian Society for Parasitology
8: 83-84
Votýpka J.,
Svobodová M., Voříšek P., Peške L., Lacina D., Volf P. (1998): Blood parasites
and haematophagous insects of raptors (Falconiformes) in the Czech Republic.
Bulletin of the Scandinavian Society for Parasitology 8: 87
Svobodová M., Votýpka J., Voříšek P. (1995): Frenkelia spp. in the populations of their final and intermediate hosts. European J. Protistol. 31: 467. Abstract from the 2nd Eur. Congress of Protistology and 8th Eur. Conference on Ciliate Biology, 21-26 July, Clermont-Ferrand, France